|
Brain models in 3D
Brain models in 3Dlabor&more in conversation with Dr Madeline Lancaster, winner of the Eppendorf Award for Young European Investigators 2014US research scientist Dr Madeline A. Lancaster (Marie Curie Postdoctoral Research Fellow, Juergen Knoblich lab, Institute for Molecular Biotechnology (IMBA) at the Austrian Academy of Sciences in Vienna) has been presented with the Eppendorf Award for Young European Investigators 2014 in recognition of her outstanding achievements in research. Worth €15,000, this highly prestigious international award has been presented annually since 1995 to research scientists working in Europe aged 35 and under. Prizewinners are chosen by an independent jury chaired by Prof. Reinhard Jahn (Max Planck Institute for Biophysical Chemistry, Göttingen). Madeline Lancaster’s recent work attracted considerable media attention far outside her specialised scientific field, since her pioneering research has now made it possible to model human brain structures in a three-dimensional stem cell culture. Madeline Lancaster was able to demonstrate how these “cerebral organoids” can be utilised to reproduce brain conditions resulting from developmental disorders in vitro. labor&more: Dr Lancaster, we’d first like to congratulate you on your receipt of this award. And we can’t deny that we’re particularly pleased about your success, since we highlighted the potential of 3D cultures in our cover story back at the beginning of the year. In his article “From 2D to 3D: A technology comes of age“ (L&M 2/14), labor&more’s Scientific Advisor Professor Paul G. Layer discussed the implications of your work as recently published in Nature (Lancaster et al., 2013, DOI: 10.1038/nature12517). In your work at the Knoblich lab, you were able to grow “mini-brains“ from stem cells for the first time – causing a worldwide sensation. What does this achievement mean to you personally? Dr Madeline Lancaster: It’s a great honour. I must admit that we had not been expecting such a large-scale reaction to our work at all, and I think the response has been simply amazing. We have been approached by researchers from a huge range of disciplines and have even received enquiries from members of the public who were simply fascinated by the potential of our work. For someone working in scientific research, I think it’s the highest accolade of all when your work is recognised by the general public and when people start becoming enthusiastic about science. In a world first, the cerebral organoids you developed at IMBA have made it possible to mimic structures of probably the most complex organ that nature has ever produced, in its early stages of development. Which regions of the brain do they represent? The mini-brains we have developed can on average reproduce the formation of brain structures until up to the ninth week of pregnancy, and have the astonishing faculty of being able to assume the identity of a range of different brain regions. These regions include the cerebral cortex – and thus the largest part of the human brain – the retina, the hippocampus, which is responsible for learning and memory, and the plexus choroideus, which is the region that is responsible for the production of cerebrospinal fluid. These various regions are all regions that are critical for the brain’s proper function and can be affected by a range of brain disorders. We can now use the cerebral organoids to investigate these disorders.
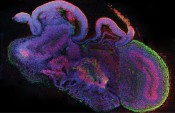
Cross-section of a complete cerebral organoid with various brain regions. Cells are shown in blue, neuronal stem cells in red and neurons in green. Photo: © IMBA/Madeline A. Lancaster
Your work uses induced pluripotent stem cells (iPS cells), whose discovery by Japanese researcher Shinya Yamanaka was recognised by a Nobel Prize in Medicine in 2012. What role does this type of cell play in organ culture and how do you utilise iPS technology in your studies? The method used to develop mini-brains starts with iPS cells. Essentially, these are mature cells that have been reprogrammed back to the state at which they were minimally differentiated. In terms of organoid development, the advantage of using such cells is that iPS cells can also be harvested from patients with brain diseases, so as to create mini-brains that reproduce these disorders in vitro. There’s also a potential therapeutic use for iPS cells in the repair or replacement of diseased organs: the advantage here is that they wouldn’t be rejected by the individual, since they originate from the same patient being treated. Although it led something of a shadowy existence at first, three-dimensional cell culture technology has been in use for decades, and has now been placed centre-stage by your work. What potential does spheroid technology have for your work, and what are its limits? As you say, three-dimensional cultures have actually been in use for many years now. Accordingly, our work has also been influenced by a large number of studies that have shown how some cells – such as epithelial cells – can form highly complex structures if they are permitted to grow in the right culture and in three dimensions. I think that our work will continue to be influenced by the work of a great many other labs in which research teams have also recognised the enormous potential offered by 3D systems. There are of course limits to this work, and this kind of system will never enable us to reconstruct the context of a complete organism in vitro. As one example, these 3D systems of course lack the blood vessels that would normally be present in a complete organ in vivo. You have deployed the cerebral organoids as a model for the simulation of microcephalies, i.e. brain development disorders that lead to a significantly smaller brain and associated intellectual disabilities. What have you discovered here? This is where the potential of iPS technology has really come into its own. We have been able to present microcephaly in model form by utilising iPS cells from patients with this disorder. We observed a truly surprising reduction in the size of the organoid, corresponding to the reduction in brain size seen in the respective patient. Further investigation revealed that this is conditional on a premature transition of neuronal stem cells to the production of neurons: these cells become exhausted too soon, ultimately resulting in fewer neurons being produced.
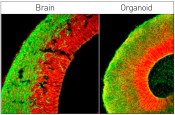
Figure showing a side-by-side comparison of a cerebral organoid (right) and the developing brain of a mouse (left). Both figures show neuronal stem cells in red and neurons in green. Photo: © IMBA/Marko Repic and Madeline A. Lancaster
Your work has also triggered criticism arising from concerns about brains created in the laboratory. What is your position on this topic? I think that most of the criticism directed at the concept of growing a brain in a Petri dish won’t really present much of a problem. What we’ve created is a piece of brain tissue with no interconnections and no sensation. Without these elements, it’s impossible for functional circuits to form: in my opinion, there’s therefore no real danger of this technique being used to create a brain capable of thought. And that’s also something that we’re not even remotely interested in pursuing, since it would be certain to raise some serious ethical objections. My research work focuses instead on understanding aspects of the brain in its early development and the creation of neurons in particular. What would be your advice to your younger colleagues who are about to embark on a career in scientific research? I firmly believe that students considered in pursuing scientific research as a profession should never forget the reasons why they decided to be a research scientist, even at later stages in their career. I’ve always been an inquisitive person by nature and, as I see it, science continues to pose such an amazing number of interesting questions that are worthy of investigation. Time and again, I’m fascinated by some new angle or other, and I take the liberty of simply pursuing this approach, wherever it may take me. I think it’s important to retain that child-like sense of wonder. Thank you for the interview, and we wish you every success for the future! (Interview: Claudia Schiller) Madeline A. Lancaster // studied biochemistry at Occidental College in Los Angeles, graduating with a B.A. in 2004 // Undergraduate research at institutions including the Department of Chemistry at the University of Utah followed in 2002–2004 // After receiving her doctorate in biomedical sciences from the Howard Hughes Medical Institute at the University of California in San Diego in 2010, she joined Juergen Knoblich’s team at IMBA (Vienna) as a Marie Curie Post-Doc Fellow // has published numerous articles in prestigious academic journals // She has most recently led research into the development of three-dimensional brain models, for which she was awarded the Eppendorf Award for Young European Investigators 2014 |
L&M int. 3 / 2014
Free download here: download here Read more articles online |