|
Scientist
>
Martina Stützle
>
How biopharmaceutical drug substances use the nose as a pathway to the brain
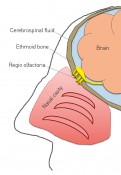
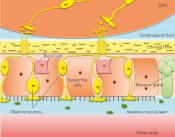
How biopharmaceutical drug substances use the nose as a pathway to the brainBehind the doorman’s backAccording to WHO estimates, over 1 billion people worldwide suffer from disorders of the central nervous system (CNS). Some of the most familiar are Alzheimer’s, Parkinson’s and multiple sclerosis. Although they have long been the focus of research, effective pharmacotherapies are still unavailable for many of these diseases. What is it about CNS disorders that makes it especially difficult to develop drug products that target pathophysiological mechanisms – so as to retard, halt, change or even reverse them? What new solution strategies are available in this field of research? The first major obstacle to the development of new active ingredients for the treatment of CNS disorders is presented by the pathophysiological mechanisms that underlie the various diseases. Mechanisms for CNS disorders are often complex and in many cases (such as Alzheimer’s) remain inadequately elucidated to date. There are, however, several highly promising candidate compounds such as insulin that significantly improve cognitive performance for Alzheimer’s in animal models. To be able to assess these potential drug substances in terms of their use in treating human patients in clinical trials, we need a practicable system that facilitates the safe transport of these candidate compounds in their biologically active form to their site of action. Strict exclusivity Depending on drug substance structure, this can often be a difficult undertaking, however: just like a club with VIP access, the human brain/CNS is an extremely exclusive location. To avoid this physiologically sensitive organ being affected by undue turmoil and general commotion, the body uses a careful process of selection to decide which molecules are granted access and which have to “wait outside”. The bouncer employed by this exclusive club is the blood-brain barrier (BBB). This is a physiological barrier between the circulating blood and the central nervous system. As a doorman, the BBB has the job of letting in “staff” (proteins such as transferrin) and “paying guests” (nutrients such as glucose) while escorting “patrons who’ve had enough for today” (degradation products such as CO2) out of the brain. All of this keeps the brain’s VIP bouncer very busy. While its colleagues (the capillary tubes at the body’s periphery) are constructed of endothelial cells equipped with openings (described as being “fenestrated”) and intracellular gaps – thus permitting them to allow dissolved and suspended molecules to pass much more easily – the blood-brain barrier consists of a continuous epithelium that is especially tightly sealed. This continuous epithelium presents an impenetrable barrier to most of the molecules transported in the bloodstream. For selected molecules, specialized transport proteins enable an exchange of substances between blood and the CNS. For a candidate compound who is new in town – and who therefore doesn’t have a transporter to call on – it is very difficult to gain access to the exclusive CNS VIP Club. But it’s not impossible. Small, lightweight molecules – the “size zero models” among molecules – in particular have a good chance of slipping by, especially if they (unlike their human counterparts) are lipophilic, i.e. easily dissolved in fats (e.g. antidepressants). Big and heavy molecules that are hydrophilic, i.e. easily dissolved in water, have little chance of making it past the bouncers. And in cases where we can see from afar that these big molecules carry a charge, their chance of being waved through by the doormen is drastically reduced. This state of affairs is especially frustrating when we recall how many new and innovative drug substances are being produced in the field of biopharmaceuticals alone. Products manufactured using biotechnology include, first and foremost, therapeutic proteins such as hormones (e.g. insulin), growth factors (e.g. erythropoietin, “epo”) and therapeutic antibodies. Of these, therapeutic antibodies are now a primary focus in drug development: as a key element of the immune system, they exhibit high-affinity binding – the “lock-and-key” principle – to pathogens, for example, and thus help to eliminate them. The nose as a backdoor Since biotechnological techniques allow us to design the structure of antibodies so that they can specifically recognize almost any molecule, they have the potential to offer targeted therapies for a wide variety of disease mechanisms while causing few side effects on account of their specificity. The problem with these therapeutic proteins is that they belong to the group of macromolecules for which the blood-brain barrier presents an insurmountable obstacle. Yet the obstacle can be bypassed instead of tackled head-on. Alongside its front door, the CNS VIP Club also has a kind of rear entrance, namely the nose – or the regio olfactoria, to be more precise (Fig. 1). This region is located right at the top of the nasal cavity. At this location, the brain and the (cerebrospinal) fluid that surrounds it is separated from the outside world by only a single bone (the ethmoid bone) and a few layers of cells (the olfactory epithelium) (Fig. 2). Since the ethmoid bone is pervaded by nerve fibers, this barrier can be overcome by a wide variety of drug substances, including therapeutic proteins. This has already been demonstrated by several groups of researchers. The research group in the Institute of Pharmaceutical Biotechnology at Biberach University of Applied Sciences, working together with Prof. Mavoungou and Prof. Schafmeister under the direction of Prof. Katharina Schindowski, has set itself the task of developing an intranasal drug delivery system that is capable of introducing drug substances in their biologically active form and with demonstrable kinetics into the brain via the regio olfactoria. The aim of this research project is to develop a system that enables the safe transportation of various active ingredients targeting disorders of the CNS to their site of action, and thus open the (back) door to the treatment of a wide range of illnesses. One well-known intranasal drug delivery system is the nasal spray, which turns drug substances into a fine mist, allowing them to penetrate deep into the nasal cavity as gas-borne aerosols. While this system works brilliantly for the small chemical molecules intended to reduce inflammation in the nasal mucosa during colds or allergy attacks, for example, it cannot be adapted to drug substance heavyweights like proteins quite so easily – since the rear entrance to the CNS VIP Club is tucked away on the roof of the nose.
Fig. 1: Section through the human skull, illustrating the regio olfactoria
Fig. 2: Structure of the olfactory epithelium and the regio olfactoria
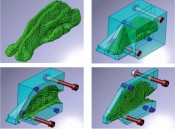
First simulate, then formulate Such particle flows through the nasal cavity are now being simulated in collaboration with the University of Ulm and Ulm University of Applied Sciences. The simulation data is intended to produce key findings such as the optimum particle size, for example. The simulations show that very small aerosol particles must be generated if we intend to penetrate into the regio olfactoria. This simulated experiment was repeated in the lab with a 3D printed model of the human nasal cavity: the lab experiment confirmed the finding (Figs. 3 and 4). This tells us that the rear entrance is not merely well-hidden: the drug substances must also be packed in vehicles for transport that are small enough to navigate the arduous path to the back door.
Fig. 3: Section through a 3D print of the nasal cavity
Fig. 4: Computer simulation of the human nasal cavity (Rolf Pfäffle, Fa. Beiter)
The particle size of such transport vehicles can be controlled with a range of techniques for atomizing the solution or altering its viscosity. Alongside the familiar pump-action nasal sprays, aerosols can also be generated with ultrasound, propellants or vibrations. To create an aerosol, energy is introduced into the medium we want to turn into a mist. Unfortunately, this procedure is sub-optimal for highly sensitive protein substances, since the force required often destroys the crucial spatial structures that the proteins need to function. By modifying the formulation (i.e. the liquid in which the proteins are dissolved), however, we have managed to identify formulations for a range of proteins that are appropriate for protecting the proteins from destructive forces during the process of aerosol generation. Our next goal is now to develop a system that allows a therapeutic drug substance to reach sufficient concentrations at the site of the regio olfactoria and within the CNS. A number of obstacles must still be overcome. In this context, one important goal for the intranasal application of therapeutic proteins is to ensure their release in a way that does not activate the immune cells present in the nasal mucosa. This would effectively vaccinate the person against the drug substance administered. If you want to get into the CNS VIP Club by the back door, it’s best to be quiet so as not to alert the security guys. There’s still a lot to do – but anyone wanting a seat in the CNS VIP Lounge has to work hard to earn it. Picture: © istockphoto.com| Chris Schmidt |
L&M int. 4 / 2015
Free download here: download here The Authors:Read more articles online |