|
A new application for microscopy
A new application for microscopy
Non-Invasive Cancer Diagnosis
Cancer has been a part of human life from our earliest days. In research conducted by Aachen-based oncologist Dr Leo Habets, a non-invasive, microscope-based diagnostic procedure has the potential to revolutionise research and progress in understanding the circumstances of the disease, so as to develop new therapeutic approaches. Microscope-based high-content screening systems in basic clinical research: can a non-invasive procedure replace the biopsy?
A 2,000-year quest
Hippocrates, the founding father of modern medicine, described growths whose blood vessels spread in all directions and were arranged like the legs of a crab. Once translated to the Latin word for crab, “cancer”, the disease now had its modern name. Yet progress was slow for both diagnosis and treatment. For two long millennia, cancer was believed to result from a surfeit of “black bile” (from the Greek “melancholia”). In the absence of any empirical data, metaphysics long reigned supreme. Today, while “black bile” won't be found in any diagnosis, many of the mechanisms by which cancer cells develop and propagate still remain shrouded in mystery. As one example, researchers have seen how cancer patients will initially respond well to modern monoclonal antibody treatments, yet some patients then suffer recurrences, relapses with new metastases, even though the disease appeared to be already beaten. Why – and how – do these new metastases occur? Are there warning signs or early indicators that hint at the danger?
Empirical basic clinical research
Aachen-based oncologist Dr Leo Habets has been studying these questions, examining samples from cancer patients on a day-to-day basis in his lab in Aachen, supported by funding from METARES – the “Society for the Promotion of Cancer Diagnosis and Treatment at the Micrometastasis Stage”. His work focuses on monitoring the success of treatment regimens for breast cancer patients. The approach he follows is empirical basic clinical research, which identifies indicators for metastases, as well as their propagation routes and mechanisms. “Our lab has studied samples from around 16,000 cancer patients,” explains Dr Habets. “And we add 16 new datasets every day, on average.” Even to the untrained eye, Habets’ workplace would seem rather unusual, since refrigerators full of tissue samples – the predominant feature of this kind of lab – are entirely absent. Apart from blood analyser units, only a few optical microscopes meet the eye. “What you see here,” he says, pointing to a microwell plate on a microscope’s specimen stage, “is the modern approach to tissue sampling: 20 millilitres of patient blood.”
Biopsy as standard – “liquid biopsy” as the alternative
Previously, cancer diagnosticians were reliant on the biopsy, i.e. the surgical removal and analysis of tissue from a patient. Nor is it merely the case that the operation is complex, expensive and fairly unpleasant for the patient concerned. The biopsy itself is now suspected of propagating tumour cells in the body. Blood tests certainly seem to indicate that the blood tumour cell count rises after tissue piercing for biopsies. A less drastic method therefore seems to be called for. “With our procedure, we can largely eliminate patient stress,” states Dr Habets. “Instead of surgery, patients merely need to have blood drawn – and we need just 20 millilitres.”
“Liquid biopsy” is what researchers are calling this new diagnostic method, which has the potential to revolutionise cancer diagnosis. “The procedure offers advantages at all stages of cancer diagnosis and treatment monitoring,” says Dr Habets. “We only have to look in the microscope to see whether or not a treatment is working. Non-invasive diagnosis even makes it possible to test drugs against patient blood before administering them. This enables the identification of potentially effective substances without inconveniencing the patient.”
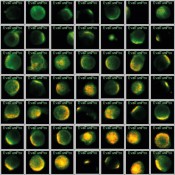
Fig.1 Maintrac procedure for cancer cell profiling with the Olympus Scan^R system. Detection of oncogenic molecular labels HER2 (orange) on EpCAM-positive cells (green).
The dangers of circulating epithelial tumour cells
“The method we use was first developed by Professor Katarina Pachmann at the University of Jena,” explains Dr Habets. “Her Maintrac procedure is extremely effective at identifying circulating epithelial tumour cells (CETCs) in blood. Their proliferation in blood is a clear warning sign. The underlying hardware is a Scan^R screening station from Olympus, running Scan^R software for data acquisition and analysis, which was specially developed by Olympus for this procedure. We use fluorescent dyes to label the cell types and cells of interest to us with antibodies.”
The differentiated display of the fluorescent dyes is a major advantage offered by the system. Cells that display all dyes as spots of colour are of particular interest. As one example, Figure 1 shows CETCs that exhibit specific oncogene characteristics due to the HER2 and EpCAM markers. Habets explains: “As you can see, some cells take up the fluorescent dye and show saturation over a wide area, while some have only spots of colour. Others have diffuse colouring or lack colours. As our data repository grows, we constantly improve the precision of our statements about the correlation between the occurrence of certain cell types and antibodies with cancer treatments and disease progression. All of the data are analysed in software, stored as image files and accessible from the database for later comparative studies.”
Fig.2 Olympus Scan^R high-content screening system. The modular all-in-one solution forms the basis for the Maintrac procedure deployed.
Scan^R: a modular high-content screening solution
The Scan^R system as deployed by Dr Habets combines the modular and flexible approach of a microscope-based system with ability to meet the requirements of screening applications in terms of automation, speed, throughput, reliability and reproducibility (Fig.2). The system is equally capable of handling both fixed and living cells. This makes the system – developed by Olympus in close collaboration with the European Molecular Biology Laboratory (EMBL) – suitable for use in a wide range of high-content screening applications. Results achieved include multiparameters and functional data about the interaction of a substance with the target or other cell components – such as absorption, permeability, selectivity, specificity or substance metabolism. Dr Habets: “The entire system is managed via a user interface which is for the most part self-explanatory. Users have access to all of the image acquisition and image analysis parameters.” The images are acquired with multiple dimensions (X, Y, Z, t), which can be evaluated subsequently with the aid of the integrated analysis system. “The options available to users in terms of image and data analysis are effectively unlimited,” says Habets. The procedural approach taken by the Scan^R system's analysis module is oriented on cytometry, which enables the analysis of a large volume of multidimensional datasets. This sort of analysis is applied in infection biology, for example, to gain new insights in relation to infectious diseases. As two examples, the Scan^R system has previously been used in work studying Chlamydia bacteria and gastrointestinal viruses. While Chlamydia bacteria predominantly affect Africa – where they can cause blindness in many cases – gastrointestinal viruses are also widespread in Europe. Across Europe, 50?% of its inhabitants are host to such viruses, which are suspected of causing cancer. Other fields of application for the system include diabetes research and the investigation of disorders such as coronary heart disease or atherosclerosis – each at the molecular level. Or – as in the case of Dr Habets' work – cancer.

Fig.3 Visualisation of metastasis risk indicators with the aid of the Maintrac procedure. Detection of CD44 antigen markets (blue) on CETC with the aid of fluorescently labelled antibodies.
Extraction guaranteed
The Olympus system currently records up to five fluorescent tumour markers in parallel per scan. For each patient, this means some 10,000 blood cells are recorded at maximum resolution using specialised auto-focus routines. Such a precise quantitative analysis is ultimately dependent on the use of a highly stabilised fluorescent light source. The analysis software setup is based on the scatter plot approach used in flow cytometry, and enables the extraction of cell populations on the basis of parameters related to both intensity and morphological features. From those 10,000 blood cells, this enables the detection of a tumour-relevant population of as few as 1–50 cells, which can be displayed directly as an image gallery. Importantly, the Scan^R software offers bidirectional linking between the data point and the individual image of the cell in question, which enables interactive assessment and manipulation of the analysis.
Vast volumes of data
The datasets involved are tremendous: every month, the research lab fills a terabyte-sized hard drive with its data. “We’d be lost without software-driven data management,” confesses Dr Habets. The fees charged for testing are moderate, however: “Our going rate for the identification of three antigens per patient is about 10 euros. That's incomparably cheaper than similar procedures.” For the moment, analysis work with the Scan^R system is confined exclusively to basic research: in the future, it could help to develop or establish diagnostic procedures.
New insights, new questions
Within oncology, there is growing demand for affordable methods in basic clinical research. For every new insight, new questions arise. Tumour cells are usually epithelial cells, i.e. cells from the surface layers of tissue complexes. If cells taken from a breast cancer patient after “successful” treatment for cancer can be shown to be circulating epithelial tumour cells (CETCs), for example, this indicates the cells have survived the therapeutic bombardment unharmed in a kind of “hibernation”. Alongside this survival, it's also a mystery why they are in the blood system in the first place. As epithelial cells, their path into this system should technically be blocked. “In a sense, they change their ‘identity’,” explains Dr Habets. “They undergo epithelial–mesenchymal transition (EMT), penetrate the blood vessels, roam about the body and can then metastasise. The Maintrac procedure gives us a reliable technique for identifying them in blood (fig.?3). Our next task is to explore the treatment options that are most effective in hampering EMT.”
Figs. 1 and 3 with kind permission of Dr Leo Habets.
Photo: © panthermedia.net | Christos Georghiou
|