|
L&Mint-int-3-2015
>
Using functional polymers in porous structures for transport control
Using functional polymers in porous structures for transport controlSwitchable nanochannelsCan molecular transport be controlled by nanoscale pores? In both human technology and Nature, many transport and separation processes are based on pores and porous materials. If transport needs to be time-controlled and separation based not merely on size, it becomes necessary to combine pores of a certain size with switchable chemical functions or polymers. In Nature, molecular transport processes are based on the interplay of structure and function at the nanoscopic scale (1nm= 1x10-9 m). Examples include ion transport through membranes with the help of ion channels and water transport through pores such as aquaporin. Transport processes can also be selective, switched or unidirectional. From a technological perspective, porous structures are important in separation processes or sensor systems, for example. Here, there is increasing interest in miniaturizing sensors and isolating them from outside factors such as pressure. Such work depends on the ability to understand and control transport processes at the micro- and nanoscale. One fascinating approach to understanding and controlling transport processes is offered by the combination of relatively stable, porous ceramic structures and switchable functional polymers. Porous films Mesoporous materials have pores with a diameter of 2 to 50nm and can be made from materials such as silica – i.e. glass (Fig.1). Methods of manufacturing such mesoporous films include the sol-gel process and evaporation-induced self-assembly, for example [1, 2]. In another procedure, block copolymer micelles are used as a template: these are burnt out following membrane separation, leaving behind an ordered, porous silica structure with a smooth surface (Fig.1). This procedure for manufacturing mesoporous films has been known of since 1999 [2] and permits the configuration of pore diameter, pore arrangement and pore connectivity. To date, these kinds of films have been produced from a variety of materials and for a wide range of potential applications. Examples include porous semiconductors for solar cells, porous glass for separation methods and porous support materials for catalysts [3]. The functionalization of mesoporous walls can be achieved either by binding organic functions after film manufacture (post-grafting) or by the addition of functionalized educts during the film production process (co-condensation). An advantage of this in-situ functionalization is the homogenous distribution of organic functions. The disadvantage is that the porous structure obtained may also be influenced. The organic functions so introduced can subsequently be used for the binding of switchable polymers.
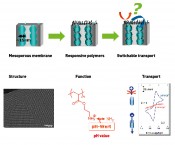
Fig. 1: Schematic diagram of the combination of a mesoporous structure with functional polymers for the switching of transport processes. An electron micrograph of a mesoporous film is shown below (left), as well as a cyclic voltammogram, that illustrates the switching of ionic pore accessibility by way of example (right).
Fig. 2: Potential stimuli-responsive polymers for transport control in combination with ceramic mesopores.
Polymer functionalization of nanopores The polymer functionalization of these porous membranes is particularly interesting: due to the monomers from which their chains are constructed, polymers possess a specific chemical function plus additional characteristics determined by their chain structure. One example is a stimulus-response behavior, for example. There are polymers that vary their degree of swelling (their expansion in a solvent) dramatically in response to slight changes in temperature. The binding of polymers to a surface also changes the properties of that surface – such as wettability and the surface charge, for example. The combination of both building blocks – nanoscale pores and functional polymers – results in the creation of a new field of research, which has recorded a huge growth in popularity over the last few years [4, 5]. Another aspect of polymer functionalization is the choice of "grafting onto" or "grafting from" – i.e. a polymerization reaction that is initiated on the surface itself. In the case of grafting onto, an entire polymer chain must diffuse onto its later support site: for large polymers and small pores, this permits the targeted functionalization of the external surface, for example. Since surfaces in mesoporous materials (pore walls) are juxtaposed at distances of only a few nanometers, surface forces and mesopore accessibility play a decisive role in polymer functionalization, in determining polymer properties in pores and for the control of transport processes by such pores. One example is the effect of pore size on chain growth during a polymerization reaction. If pores are smaller than 10?nm in diameter, then the probability increases that radical polymerization reactions break off relatively quickly. One reason for this happening is that the radicals responsible for chain growth react with one another and not with a new monomer [6, 7]. Alongside the polymer functionalization itself, polymer behavior is another aspect influenced by the confined spaces available in nanoscale pores: as one example, we know that the pKa value of polymers can be shifted to more extreme pH values [8]. Theoretical calculations have also shown that, as a function of chain length and pore size, polymers can collapse either towards the pore wall or even to the pore center, depending on whether an increase in polymer-polymer association or the entropic losses of chain stretching are favored. Polymer distribution also has a role to play [9]. Understanding this polymer behavior is a basic precondition for the targeted control of molecular transport processes by pores of this kind. Controlling molecular transport in nanopores In this field of research, many examples over the last few years have demonstrated that switchable mesopore accessibility is possible for charged molecules [4, 10]. This research has investigated a range of polymers and switching stimuli (fig.2). A switching of the polymer charge – and thus of mesopore accessibility – has been achieved by altering the pH value from acidic to basic, by complexation reactions involving multivalent ions, by light, by temperature and, just recently, by redox reactions (Fig.2) [4, 13]. The common principle remains the combination of pore size and polymer chain length. If the polymer is charged using a stimulus, the sphere of influence for its electrostatic charge is described by the Debye screening length. If pore size is on the nanoscopic scale, then Debye screening length and pore size are comparable – even for relatively high concentrations of ions. Accordingly, the polymer bound to the pore wall acts as a "bouncer" for small charged molecules: the pore will now no longer filter just by size but also by charge. Molecules with the same charge cannot force their way into the pores, while molecules carrying an opposite charge can be concentrated. While the control of molecular transport by charge-switchable polymers is now possible, many challenges remain to be solved in this field. Questions have still to be answered not only regarding the control of polymerization reactions and thus pore charge, but also in relation to the opportunities and limits to local control in nanoscale functionalization. Initial studies show that an incremental configuration of polymer quantity and thus charge leads to an incremental control of the ionic transport [7, 11]. Within certain limits, the quantity of a permanently-charged polymer can be controlled via the polymerization reaction time, leading to a variation in the ionic pore accessibility – from molecule exclusion through to molecule concentration. Knowledge of such transport processes can also be deployed in a synthetic context: as one example, catalysts can be bound to these pores and starting materials for nanoparticles concentrated via surface charging, thus creating catalytically or optically active nanoparticles in porous membranes. The enclosure of metallic nanoparticles of gold or platinum leads to catalytically active pores, for example. The ability to make such mesopore transport processes switchable is also being investigated in medical applications. One example is the use of porous nanoparticles as carrier systems for the local release of drugs within tumor cells [12]. The combination of ceramic mesoporous materials with functional, switchable polymers is therefore not confined to the investigation and clarification of molecular transport processes. Instead, it may well prove useful in the long term for other areas of research and potential applications in fields such as catalysis, separation, sensors and medicine.
Bibliography picture: © panthermedia.net | koya79 |
L&M int. 3 / 2015
Free download here: download here The Author:Read more articles online |